Options & accessories - Bertin Medical Waste

Options & accessories
To complete its range of ultra-compact solutions, Bertin Medical Waste offers options and accessories that can be combined with the machines to provide customers around the world with personalized turnkey solutions.
Tipper machine
Bertin Medical Waste has developed different types of container tipper machines for automatic loading of biomedical waste into the Sterilwave vessel.
These loading methods using a tipping motion allow safe handling by the operator and save time during the loading phase. Risks of contamination are therefore reduced and productivity increased. We offer a 240-litre container tipper machine for Sterilwave 250, and a 360-litre version for Sterilwave 440. We can supply a 1,000-litre version upon request. Other sizes can also be adapted to fit our equipment.
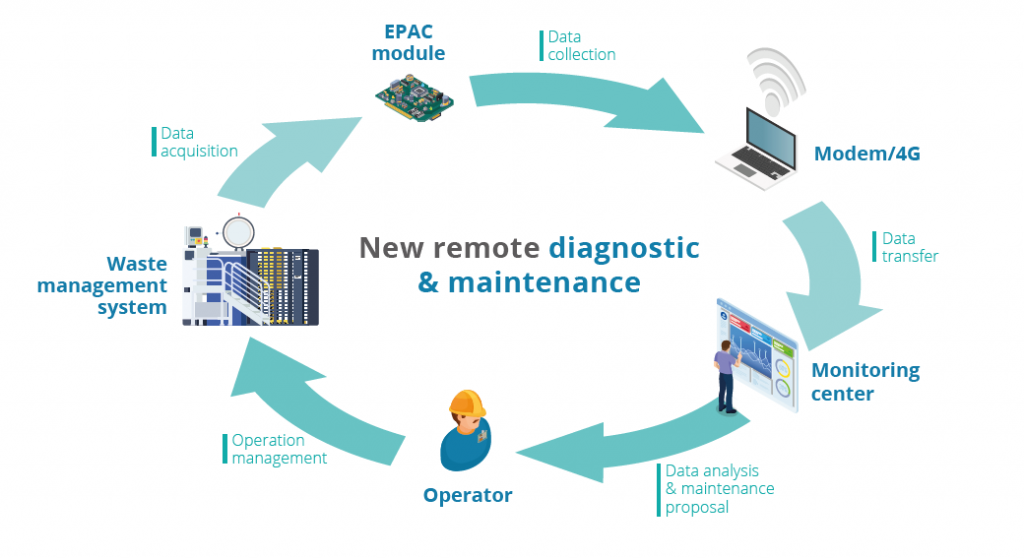
Biomedical waste treatment tracking
The Sterilwave systems are all fitted with a machine controller which records all the parameters from each biomedical waste treatment cycle. At the end of each cycle, the operator retrieves a printed ticket showing the essential information (weight of input waste/time cycle was started/success of decontamination process).
The system’s production can also be monitored by consulting the cycle and results log which can be retrieved via an IP connection or SD memory card. For maintenance purposes, all measurements and parameters can be retrieved in this way in order to analyse and identify any faults. Bertin Medical Waste also offers tailor-made solutions, adapted to your needs, for the tracking of your biomedical waste treatment.
Microbiological inactivation monitoring kit
Operators are required to perform regular checks of the effectiveness of microbiological inactivation in the Sterilwave systems. In fact, we recommend carrying out these tests at various intervals in accordance with current regulations or after major maintenance work, in order to verify that the system is working properly. Bertin Medical Waste has developed a bacteriological kit for this purpose which has been laboratory certified. In a simple procedure, the operator can check themselves whether the level of bacteriological inactivation is higher than 6log10 using bacillus spores. The test is performed during a machine cycle and results can be obtained within 48 to 72 hours. This ensures the full traceability of biohazardous waste treatment.
Odour & vapour management
the Sterilwave’s microwave technology produces only dry final waste material. As a result, odours linked to the treatment of biomedical waste are minimised when compared to other procedures. To improve the operators’ environment even further, Bertin Medical Waste has developed two solutions for reducing odours from biomedical waste during the Sterilwave process. The first consists of using a gel which masks the odours. This is applied manually by the operator during each cycle (about 10 grams per cycle). The second consists of installing an ozometer which catalyses the odour molecules in the fume evacuation duct. The ozone is produced from the surrounding air and complies with current environmental restrictions. This device is managed directly by the machine controller and is much more effective at reducing odours.
During the heating and sterilisation phase, the microwave process reduces the weight of the biomedical waste by removing its water content. It then converts it into vapour which is evacuated via a HEPA bacteriological filter. Bertin Medical Waste offers different evacuation solutions for this vapour, adapted to each institution taking into account each building’s particular constraints. If the evacuation is at a distance of several metres, Bertin Medical Waste offers CMV type extractors which can eject any water vapour into the relevant site’s areas suited to this purpose. In collaboration with our technical services department, we can provide your organisation with custom-made support which can help define the best solution for optimising vapour flow.
ANY QUESTIONS? CONTACT AN EXPERT
Please fill in this form if you wish to contact our sales team or receive further information.